MRI-guided SABR boost when brachytherapy is not feasible: emerging evidence and a real-world clinical case
Introduction
A proportion of patients with locally advanced cervical cancer are unable to undergo brachytherapy due to tumour extent, anatomical constraints, or medical comorbidities. In these situations, delivering an adequate tumour dose while maintaining acceptable exposure to surrounding organs at risk represents a significant clinical challenge.
Concurrent chemoradiotherapy followed by a brachytherapy boost remains the established standard of care for patients treated with curative intent.1 However, when brachytherapy is not feasible, alternative boost strategies must be considered.
Conventional external beam radiotherapy boost is often limited by prior radiation exposure to surrounding tissues and may not achieve the high central tumour doses associated with optimal outcomes. Advances in radiotherapy delivery have therefore created new opportunities to address this gap.
In selected patients, MRI-guided stereotactic ablative radiotherapy (SABR) delivered on an MR Linac may enable delivery of a high-dose boost with improved soft-tissue visualisation and online adaptive planning. Emerging clinical evidence suggests that this approach may represent a feasible treatment option where standard boost techniques cannot be delivered.
Evidence supporting MRI-guided SABR boost
A retrospective feasibility study² evaluated MRI-guided SABR boost following pelvic chemoradiotherapy in patients with locally advanced or recurrent gynaecological cancers who were unsuitable for brachytherapy.
Ten patients (median age 56) with non-metastatic disease were treated, including recurrent cervical (n=5), primary locally advanced cervical (n=2), and recurrent vaginal cancers (n=3). Brachytherapy was not feasible due to tumour infiltration into pelvic structures, frequently involving adjacent organs such as the bladder or rectum, precluding safe applicator placement.
Patients received pelvic chemoradiotherapy (45 Gy in 25 fractions, with concurrent cisplatin in most cases), followed by MRI-guided SABR boost delivered on a 0.35 T MR Linac. The median boost dose was 21 Gy in four fractions (5.2 Gy per fraction), delivered on alternate days with online adaptive planning for all fractions.
At a median follow-up of nine months, local control was achieved in 90% of patients, with complete response in 60% and partial response in 20%. Treatment was well tolerated, with no grade ≥3 toxicities and no worsening of acute gastrointestinal or urinary effects following the SABR boost.
These findings suggest MRI-guided SABR boost is a feasible and well-tolerated option in patients who are not candidates for brachytherapy. However, as brachytherapy remains the standard of care, this approach should be considered a selective alternative rather than a replacement.
Clinical case study
The following case illustrates how MRI-guided SABR using an MR Linac was successfully implemented as a boost following chemoradiotherapy.
Case overview
A 75-year-old patient was diagnosed in early 2025 with bulky FIGO stage IIIB, grade 3 HPV-associated squamous cell carcinoma of the cervix with suspected bladder invasion.
Clinical examination revealed a large fixed cervical mass extending into the upper anterior vaginal wall. MRI demonstrated a large cervical tumour causing cervical stenosis, fluid accumulation within the uterus and indentation of the posterior bladder wall. PET-MRI confirmed a metabolically active central tumour without evidence of nodal disease.
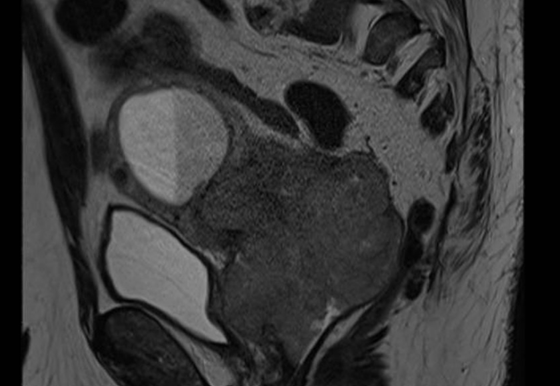
Figure 1: Baseline sagittal T2 MRI showing the presence of a large cervical tumour causing cervical stenosis, uterine fluid accumulation, and posterior bladder indentation
Brachytherapy was planned as the standard boost following chemoradiotherapy; however, two attempts at applicator placement were unsuccessful due to technical difficulties. As further dose escalation with conventional external beam radiotherapy was limited by dose constraints to surrounding organs, MRI-guided SABR delivered on the MR Linac was offered as a potential curative alternative boost strategy.
Treatment parameters
The patient began treatment with induction chemotherapy consisting of weekly carboplatin and paclitaxel, followed by pelvic chemoradiotherapy delivering 45 Gy in 25 fractions with concurrent weekly cisplatin.
Following chemoradiotherapy, the patient received 30 Gy in five fractions delivered on alternate days with a 1.5 T MR Linac, with a simultaneous integrated boost to 35 Gy delivered to the clinical target volume according to the institutional gynaecological boost protocol.
Daily adaptive planning was performed for each fraction, with only minimal optimisation required. The delivered dose closely matched the planned dose, with less than 7% variation across target and organ-at-risk parameters.
Outcomes
Clinical examination demonstrated a complete response, and MRI and PET imaging performed 12 weeks after treatment confirmed complete radiological and metabolic response with no evidence of residual disease.
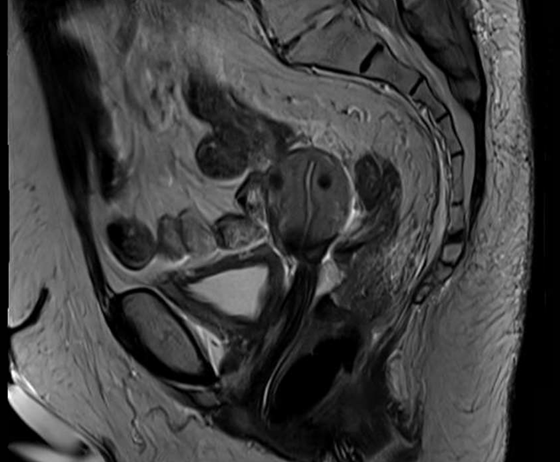
Figure 2: Sagittal T2 MRI at 12 weeks post-treatment confirming complete resolution of cervical tumour
The patient tolerated treatment well, with no increase in treatment-related toxicity following the start of MRI-guided SABR. Side effects associated with the preceding chemoradiotherapy had largely resolved by six weeks after treatment.
Patient-reported outcome measures demonstrated a substantial improvement in quality of life. Overall quality of life improved from 1 (very poor) at baseline prior to the MRI-guided boost to 7 (excellent) six weeks after treatment. Overall health improved from 4 to 7 during the same period.
Conclusion
This case demonstrates that MRI-guided SABR boost can provide a curative-intent treatment option in patients for whom brachytherapy is not feasible.
The ability to visualise soft-tissue anatomy and adapt treatment plans daily enabled accurate tumour targeting while maintaining dose constraints to surrounding organs. In this patient, MRI-guided SABR allowed successful dose escalation following chemoradiotherapy without increasing treatment-related toxicity.
Together with the findings from the study described above, this case highlights the potential role of MRI-guided SABR as an alternative boost strategy in carefully selected patients with locally advanced gynaecological cancers when brachytherapy cannot be delivered.
Refer today
To refer to one of our specialists or to find out how we can help you and your patients, contact us at the below email.
Dr Gemma Eminowicz
Consultant Clinical Oncologist
GenesisCare UK
References
1. Abu-Rustum NR et al. NCCN Guidelines® Insights: Cervical Cancer, Version 1.2024. J Natl Compr Canc Netw. 2023 Dec;21(12):1224–1233. doi: 10.6004/jnccn.2023.0062.
2. Hadi I et al. MR-guided SBRT boost for patients with locally advanced or recurrent gynecological cancers ineligible for brachytherapy: feasibility and early clinical experience. Radiat Oncol. 2022 Jan;17(1):8. doi: 10.1186/s13014-022-01981-z.
