- Healthcare Professionals
- Case studies
- Head and neck case study
Head and neck cancer case study
A case study of chemoradiotherapy following transoral surgery to treat tonsil cancer
Case study
Chemoradiotherapy following transoral surgery to treat tonsil cancer
Dr. Nicola Dallas

She has a special interest in technical radiotherapy and introduced the use of intensity modulated radiotherapy (IMRT) and now dynamic arc IMRT for the treatment of head & neck cancers to the centre.
Case presentation
A 55-year-old male presented in clinic following a chest infection and persistent swelling of the left tonsil.
A neck MRI was requested and demonstrated a 27mm x 15mm x 18mm left tonsil mass, with an associated enlarged left level 2a lymph node. A biopsy of the left tonsil confirmed p16 positive squamous cell carcinoma (SCC) and an FDG PET-CT identified the tonsil primary and associated cervical lymph node. Importantly there was no distant metastatic disease.
The diagnosis was subsequently confirmed as T2N1 p16 positive squamous cell carcinoma of the left tonsil.
The gentleman was otherwise completely fit and well, a non-smoker consuming in the range of 6-12 units of alcohol per week. He lived with his family and previously worked as a banker.
Challenges of presentation and choice of treatment
Initial discussion focused on two potential treatment plans.
Either primary chemoradiotherapy given with curative intent, or alternatively initial transoral laser surgery to remove the primary tumour, together with a neck dissection. Following MDT review the patient was referred to Prof. Stuart Winter, Consultant Head and Neck Surgeon, and the transoral laser surgery and neck dissection was agreed as the first intervention, to potentially enable a reduction in the intensity of the adjuvant treatment and subsequent toxicity.
Treatment
Post-operative assessment confirmed pT2 (30mm) pN1 (25mm) with extracapsular extension p16 positive L tonsil SCC. The case was once again reviewed in the MDT and agreed for adjuvant chemoradiotherapy.
Volumetric modulated arc therapy (VMAT) was planned 65 / 60 / 54 Gy in 301 fractions to the left oropharynx and neck, together with weekly cisplatin chemotherapy. Each radiotherapy session involved a short period – just a few minutes – on the couch and the plan was delivered with curative intent.
The patient received this course of treatment at the GenesisCare outpatient centre in Windsor with the support of the dedicated head and neck multidisciplinary team. This included weekly consultant appointments, a nurse specialist, dietitian reviews and speech and language therapist (SALT) support.
The gentleman also opted for the complimentary exercise medicine course at GenesisCare in Windsor and received a personalised prescription to support his physical and mental strength and resilience.
He also received acupuncture and reflexology through our partnership with the cancer charity, Penny Brohn UK.
Side effects and management
The patient experienced side-effects, including a sore throat and mouth. These were managed with painkillers and mouth washes, together with alterations to his diet to softer and liquid foods that were less likely to sting. Other side effects such as a dry mouth, loss of taste, skin soreness and skin desquamation were all experienced, and lots of advice given on how best to manage them. They gradually resolved over time following the completion of radiotherapy.
By 12 weeks, the skin had healed, taste was recovering and he no longer needed painkillers. Although the patient still found spicy food challenging, he was able to eat all food textures.
Results and follow-up
Close clinical follow-up continued post treatment with weekly dietitian contact and monthly consultant appointments.
An FDG PET/CT, 12-weeks following the end of treatment, demonstrated a likely complete response with some persistent post treatment inflammation visible.
Further outcome measures will be assessed through validated PROMs collected during and post treatment.
The patient’s progress will continue to be monitored for up to five years f
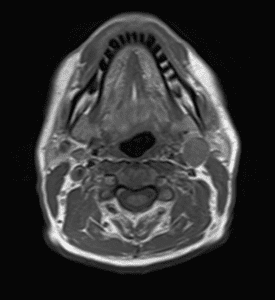
1. T1 MRI axial slice showing left level 2a lymph node from left tonsil primary.
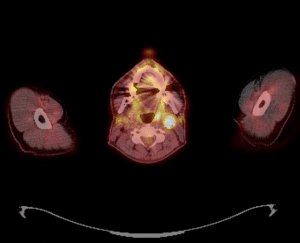
2. FDG PET-CT fused axial image showing left level 2a lymph node with left tonsil primary.