MRIdian: benefits for prostate patients
The technological benefits of prostate radiotherapy on the MRIdian.
Prostate cancer is the most common cancer in men in the UK. Around 6 out of 10 men will have localised disease at presentation, and approximately a third of these will go on to have curative radiotherapy. Radiotherapy treatment for prostate cancer has changed beyond recognition over the last 20 years, due to advances in radiotherapy technology and the machines on which we deliver treatment.
We, a group of dedicated, specialised clinicians, have now treated approximately 30 prostate cancer patients using the MRIdian. Treatment on the MRIdian allows us to confidently and precisely deliver highly targeted radiotherapy, optimising cancer kill and healthy tissue sparing, through the features outlined below.
The MRIdian planning process allows prostate delineation on a hybrid MR (magnetic resonance) scan (A). This taken alongside a planning CT (computerised tomography) scan (B) and diagnostic MR scan provides clear images for contouring the prostate. Traditionally, prostate radiotherapy has been planned by outlining the prostate on a CT scan (B), using additional information from a diagnostic MR scan.
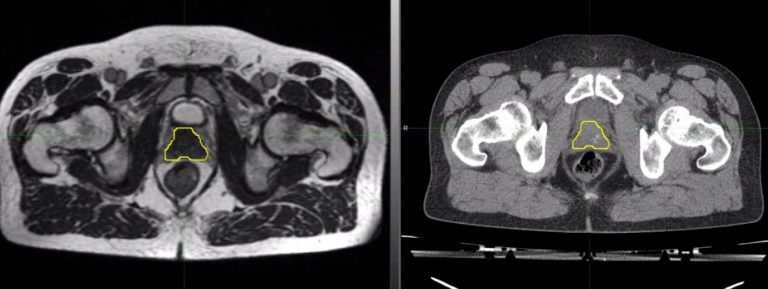
A (left image) Hybrid MR scan used for planning MRIdian -based radiotherapy, with prostate delineated (yellow contour)
B (right image) Standard Planning CT scan used for conventional radiotherapy for the same patient, with prostate delineated (yellow contour)
Radiotherapy treatment plans are produced using organ contours from a planning scan; however, these are images from one moment in time. There is internal movement of the prostate, dependent on rectal and bladder filling, even breathing. On conventional linacs (linear accelerators), imaging of the prostate is undertaken just prior to each radiotherapy treatment delivery and the treatment plan is matched to this, using as a surrogate, implanted markers in the prostate and x-rays, or a low dose CT or a combination of the two. There is often also bowel and bladder preparation prior to each treatment to reduce movement of the prostate and optimise the size and position of the rectum, bladder and bowel and reduce the risk of side effects.
Treatment delivery on the MRIdian allows daily modification of the prostate contour to allow for changes in its position or shape. It also allows assessment and modification of the rectum, bladder and bowel contours, which reduces the need for strict pre-treatment bowel and bladder preparation.
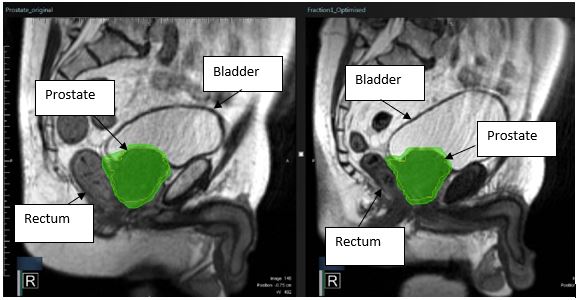
The original treatment plan is then recalculated according to what is seen on the day but also may be reoptimized, ensuring the prostate receives the dose required to maximise cancer cell kill, whilst keeping the doses to the adjacent organs within tight specified limits, reducing the risk of toxicity and side effect.
Original prostate treatment plan-prostate (yellow contour) covered by optimal, appropriately shaped, treatment dose contour (green wash).
Optimised treatment plan for first radiotherapy treatment- prostate remains covered by optimal, appropriately shaped, treatment dose, following modification for a change in prostate position due to changes in bladder and rectal size.
The MRIdian also allows continuous monitoring of the prostate during treatment delivery. Any movement of the prostate outside of a tightly defined margin results in a pause in treatment until the prostate, the target, moves back into the correct position. This gives confidence that all the planned dose is delivered to the prostate. There is no imaging of the prostate during treatment delivery with a conventional linac.
Movement of the prostate during treatment delivery due to rectal gas.
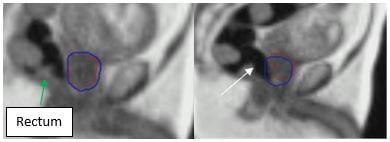
A (left image)
Treatment is delivered when prostate (red contour) is contained within a tightly defined treatment margin (blue contour).
B (right image)
Gas (or wind, white arrow) passing through the rectum, moved the prostate (red contour) out of the defined treatment margin (blue contour), ‘pausing’ treatment.
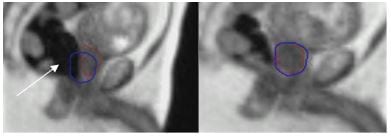
C (left image)
Gas (or wind, white arrow) passing through the rectum, moved the prostate (red contour) out of the defined treatment margin (blue contour), ‘pausing’ treatment.
D (right image)
Once the gas had passed, the treatment resumed as the prostate moved back into the treatment margin.
The prostate cancer patients we have treated on the MRIdian have spoken favourably of their experience, and the low levels of side effects during and in the weeks and months following treatment. This is in line with the available published data and compares favourably with treatment on a conventional linac.
Through the features described here, the MRIdian has allowed us to take prostate cancer radiotherapy to the next level, improving the accuracy and confidence with which treatment can be delivered to men with localised prostate cancer.
Kind regards,
Consultant Oncologist.