- Healthcare Professionals
- Oncology
- Treatments
- Radiotherapy
Volumetric Modulated Arc Therapy (VMAT)
Delivers a highly precise and personalised dose to the target area while minimising dose to adjacent organs. Significantly improves treatment outcomes for many tumour types, including breast, prostate, head and neck, skin and palliative treatments.
VMAT is the next generation of intensity-modulated (IMRT), delivering a highly conformal radiotherapy dose by changing intensity as it moves around the tumour site. This reduces treatment time as well as the overall toxicity of treatment. VMAT is used to treat many cancer types, including breast where we are pioneering the use of Partial Breast Irradiation. It is also used for palliative treatment as it preserves the opportunity for subsequent radiotherapy.
We provide VMAT to all suitable patients and use simple automation and Physics QA automation (Mobius) to create highly conformal plans for dosimetry.
Stereotactic ablative radiotherapy (SABR)
Leading providers of the most comprehensive stereotactic service in the UK
Also known as stereotactic body radiation therapy (SBRT), SABR is one of the most important advanced radiotherapy techniques currently available for the treatment of tumours such as in the lung and liver. It focuses beams of high-dose radiation at tumour sites with sub-millimetre accuracy – with the aim of achieving local control in just a few short treatments. There is a large body of evidence that SABR treatment given at the right time, to the right patient, can make a difference to outcome.[1] SABR, and stereotactic radiosurgery (SRS) for brain, are becoming the gold standard therapy for certain primary and many metastatic cancers, often replacing surgery.
GenesisCare is an established provider of specialist SABR treatment. We have invested in a network of lead centres that offers world-class technology, including sophisticated imaging, motion monitoring and the UK’s first MRIdian, for real-time adaptive planning and delivery. Our service is led by a team of expert NHS clinicians with extensive experience of this SABR modality for all tumour types and they oversee a nationwide rapid access pathway.
SABR - a powerful tool in the treatment of primary and metastatic tumours
Stereotactic ablative radiotherapy, or SABR, targets intensely powerful beams of radiation to tumours in a highly conformal treatment. It targets with the aim of destroying cancer cells with minimal damage to surrounding healthy tissues.
SABR is an extreme hypofractionated treatment that can be delivered in just one to five fractions. It has revolutionised the treatment of tumour sites by offering an evidence-based alternative to surgery and to other invasive interventional procedures such as radiofrequency ablation (RFA), microwave ablation (MWA), irreversible electroporation (IRE, ‘NanoKnife’), and cryotherapy. Numerous studies have shown that SABR is highly effective in the management of primary and metastatic tumours, in particular inoperable tumours in the pancreas and liver; secondary tumours in the liver, lymph nodes, lungs, and bones; and recurrent cancer in the pelvis after previous radiotherapy. SABR may also be an alternative to surgery for prostate cancer, depending on the tumour stage.
How SABR works
SABR is an advanced form of precision radiation therapy that exploits the principles of the radiobiology of tumour cells. The high ablative doses used in SABR not only cause lethal damage to the DNA of cancer cells (as in standard radiotherapy), but also damage the tumour stroma (the basement membrane, fibroblasts, extracellular matrix, immune cells and vasculature). The combined effect is to significantly accelerate cell death (apoptosis).[3]
Compared to conventional radiotherapy, the ability of SABR to target tumour volume and yet avoid healthy tissue with sub-millimetre accuracy is critical to outcomes of treatment. For this reason, delivery of a SABR service requires significant investment in technology and expertise, together with a robust clinical framework. This is the foundation of the GenesisCare SABR centre of excellence.
How the dose compares
SABR – dose-per-fraction of 6.7 to 10 Gy in 4 or 5 fractions with total dosage between 33.5 Gy and 50 Gy
Conventional fractionation – doses-per-fraction in the range of 1.8–2 Gy and total dosage can amount to >80 Gy with intensity-modulated radiotherapy
Hypofractionation – doses-per-fraction of 2.5 Gy and above in 20 fractions, so total is 52.5–55 Gy
The GenesisCare SABR service
GenesisCare has established a centre of excellence for SABR and stereotactic radiosurgery (SRS) led by a team of leading SABR specialists operating across five centres.
The service draws on the best practice of the world’s leading SABR treatment centres and is designed to deliver clinical excellence at each stage of the pathway. Our objective is that all eligible patients have an opportunity to benefit from SABR, with treatment starting within five days of their planning scan (or sooner for palliative patients).
Diagnostic imaging – our treatment pathway includes full diagnostics, PET/CT, MRI and CT to ensure appropriateness of patients and minimal delays in commencing treatment planning.
Referral expertise – we have refined SABR referral criteria based on the experience of our SABR specialists, published clinical studies and international best practice. All referrals are peer- reviewed by our senior SABR specialists.
Unified protocols for dose and fractionation – we use standardised dose and fractionation schedules based on tumour size and evidence-based best practice. Our dosimetrists and medical physicists calculate high-quality contouring plans, taking account of the normal tolerance of healthy tissue, organs at risk
and inter-fractional adjustments. We use a sophisticated AI planning system to calculate target volumes and all plans are peer-reviewed in multidisciplinary teams (MDTs).
High-quality imaging and set-up – we use image guided radiotherapy (IGRT) based on 3D cone beam verification immediately prior to treatment for accurate set-up. These are compared and matched with simulated X-rays taken directly from 3D CT data used for treatment planning. Our radiographers have extensive experience of CBCT interpretation and use of specialist positioning equipment to ensure sub- millimetre accuracy in set-up and minimal delay.
Superior motion management – our state- of-the-art linear accelerators integrate surface guided radiotherapy (SGRT) techniques to monitor for movements which could result in dosimetric inaccuracies. This surface data is captured in real time to minimise intrafraction variation. We also use respiratory motion management, such as breath hold, gating and beam hold. Surface data is captured at each attendance to manage interfraction variation.
Peer Review – we are committed to providing a MDT approach to the implementation of safe and evidence-based radiotherapy.
Latest-generation SABR technology – treatments are delivered with the latest- generation linear accelerators. Designed for
the technical demands of SABR, these systems incorporate IGRT and SGRT technologies. We have also introduced MR-guided radiotherapy which uses real-time MR images for truly adaptive SABR treatment.
Combined with our advanced treatment planning imaging capabilities, including 3D CT, PET/CT and MRI, we offer the most technologically advanced SABR network in the UK
MRIdian. Real-time, on-table adaptive radiotherapy - a new frontier for SABR delivery
We’re proud to be the first independent healthcare provider in the UK to introduce the MRIdian – an advanced MR linac. This world-class radiotherapy system is available at our centres in Oxford and Cromwell Hospital in London and can be accessed by patients throughout the UK. We’re offering this modality as a specialist service within our SABR network.
By combining a linear accelerator with a high definition MR scanner, MRIdian allows clinicians to see as they treat, adjusting the beam to allow for movements and anatomical changes and making truly adaptive SABR possible for the first time.
We are excited to be partnering with the University of Oxford and a team of expert SABR clinicians to build a body of evidence and put us at the forefront of exploring the possibilities for this innovative approach.
Find out more about MRIdian MR-guided radiotherapy in the MRIdian tab.
- Palma et al. Lancet 393 2051–58 (2019)
- Murray et al. Semin Radiat Oncol 27 247–55 (2017)
- Bremnes et al. J Thorac Oncol 6 1 209–217 (2011)
5# SABR for prostate cancer
5# SABR for low- to medium-risk localised prostate cancer
Following three major non-inferiority phase 3 randomised controlled trials [1,2,3] have shown its safety and efficacy, moderate hypofractionation (20 fraction) has been adopted as a standard-of-care option for localised prostate cancer.⁴,⁵ There is now a growing body of evidence in support of extreme hypofractionation – 5 fraction (5#) SABR for patients with low- to intermediate-risk (medium-risk) and selected high-risk localised prostate cancer.
We offer this innovative treatment at our centres in Bristol, Birmingham, Cambridge, Chelmsford, Elstree, Maidstone, Milton Keynes, Nottingham, Portsmouth, Windsor and Southampton on our state-of-the-art linacs and at our centres in Oxford and London using the ViewRay MRIdian MR linac. Find out more about MRIdian MR-guided radiotherapy.
Benefits to patients
We have made the 5# SABR for prostate cancer service available to offer patients an option which will provide:
- A suitable alternative to surgery – this non-surgical treatment for prostate cancer is especially relevant for patients with co-morbidities in order to avoid a hospital stay or long waiting times – particularly during the COVID-19 pandemic
- No increase in gastrointestinal or genitourinary side effects compared to 20#
- A reduction in treatment time and less interruption to the patient’s daily life
- The additional benefit of a rectal spacer to help reduce damage to surrounding healthy tissues such as the bowel, offered at no extra cost
Our aim, as always, is to deliver an exceptional service that improves outcomes for our patients.
You can download our current prostate protocol here
Evidence base
MIRAGE Trial: findings from the first phase III randomised clinical trial to assess MRI guidance vs. CT guidance in the delivery of 5# SABR
MIRAGE is a randomised clinical trial that compares MRI guidance versus CT guidance in the treatment of patients with prostate cancer receiving 5# SABR at the same radiation dose7.
Conducted at the University of California, participants were randomly allocated to receive 5# SABR via CT guidance (on the TrueBeam or Navalis Tx) or MRI guidance (on the MRIdian MR linac).
A dose of 40 Gy was delivered to the prostate in 5# using planning margins of 4mm for the CT arm and 2mm for the MRI arm.
Of the 154 patients (76 CT-arm; 78 MRI-arm) that were eligible for evaluation, the results from this study showed that:
- 24.4% of patients receiving MRI-guided 5# SABR experienced acute grade ≥2 GU toxicity, compared to 43.4% of patients in the CT-guided group (p = 0.01).
- 0.0% of patients receiving MRI-guided 5# SABR experienced acute grade ≥2 gastrointestinal (GI) toxicity, compared to 10.5% in the CT-guided group (p = 0.003).
These results suggest that using MRI guidance to deliver 5# SABR offers significant advantages over CT guidance; patients receiving MRI-guided SABR reported significantly less bowel and urinary upset, with quicker recovery times.
PACE-B: acute toxicity findings from an international, randomised, open-label phase 3, non-inferiority trial
PACE-B is a randomised trial of standard-of-care conventionally fractionated or moderately hypofractionated radiotherapy versus five-fraction stereotactic body radiotherapy for low-risk to intermediate (medium)-risk localised prostate cancer.⁶
Participants were randomly allocated (1:1) to conventionally fractionated or moderately hypofractionated radiotherapy (78 Gy in 39# over 7-8 weeks or 62 Gy in 20# over 4 weeks, respectively) or stereotactic body radiotherapy (36·25 Gy in 5# over 1–2 weeks).
Side effect data collected over the first three months after treatment, show that patients who had SABR had similar and low levels of side effects when compared to the patients who had conventionally fractionated or moderately hypofractionated radiotherapy.
The worst acute RTOG gastrointestinal toxic effect proportions were grade 2 or more severe toxic events (such as, diarrhoea, discomfort in the back passage, or changes in bowel habit) in:
- 53 (12%) of 432 patients in the conventionally fractionated or moderately hypofractionated radiotherapy group
- 43 (10%) of 415 patients in the stereotactic body radiotherapy group
N.B. this was a non-statistically significant difference.
The worst acute RTOG genitourinary toxicity proportions were grade 2 or worse toxicity (such as, needing to pass water more frequently, problems passing water or bladder incontinence) in:
- 118 (27%) of 432 patients in the conventionally fractionated or moderately hypofractionated radiotherapy group
- 96 (23%) of 415 patients in the stereotactic body radiotherapy group
N.B. this was a non-statistically significant difference
These results suggest that substantially shortening treatment courses with SABR does not increase gastrointestinal or genitourinary acute toxicity. Late toxicity and efficacy data for the PACE-B trial are awaited.
Rectal spacers and fiducial markers
As part of our imaging protocol, we include the use of fiducial markers alongside rectal spacers to improve the accuracy of treatment and reduce the effects of toxicity to surrounding healthy tissue.
The spacer is inserted between the rectum and the prostate under local anaesthetic before radiotherapy treatment begins to temporarily move the anterior rectal wall further away from the prostate. This helps reduce the amount of radiation delivered to the rectum and limits toxicity, therefore reducing bowel, urinary and sexual function related side effects. The fiducial markers are inserted at the same time, reducing the need for further procedures. Fiducial markers are an established method of improving the accuracy of treatment delivery, providing reference points for planning and monitoring treatment.
The use of rectal spacers prior to radiotherapy is recommended to reduce the potential effects of rectal and bladder toxicity for the following treatment schedules:
- Consecutive days fractionation on the MRIdian
- High-risk patients receiving an SIB to a posterior volume
The rectal spacer insertion procedure can be performed prior to or on the same day as the MR linac simulation.
For patients receiving 5# SABR on the MRIdian MR linac at our centres in Oxford or London, fiducial marker insertions are not required due to daily on treatment adaption that is made possible with real-time MR imaging.
Who can I refer?
If you wish to refer a patient for 5# SABR for primary prostate cancer on either a conventional linac or the MRIdian MR linac, the eligibility criteria are:
- Biopsy proven adenocarcinoma prostate, Gleason ≥ 6
- T-Stage: T1c-T3b
- No evidence of lymph nodes or distant metastases
- Prostate volume ≤ 90 cc
- IPSS ≤ 19
- No TURP within eight weeks
- No artificial hips (these are only suitable for treatment on the MR Linac)
- No inflammatory bowel disease
How can I refer?
If you’re an oncologist currently working with GenesisCare but are not routinely treating SABR (in private practice or in your local Trust) or have not participated in the PACE trial, we have a simple credentialing process to support you in referring and treating your prostate patients with 5# SABR. For clinicians who would prefer, eligible cases can also be referred to any credentialed clinicians to consent, plan and treat the patient before referring back to the original clinician for ongoing follow up.
The referral process and credentialing opportunity is detailed below.
Credentialing
If you have a patient who is accepted by the eMDT as eligible for the 5# prostate SABR protocol but you do not practice SABR routinely in your NHS or private practice and have not participated in the PACE trial, you can still access this treatment for your patient by following the credentialing process, stated below:
- Upon receipt of an initial linac prostate SABR referral, you will be allocated a mentor from the USAT who will deliver a prostate SABR credentialing presentation, which includes education around the prostate SABR technique, dosimetry and our protocol
- For the first three referrals, the allocated mentor will support you with providing joint review of contours and final treatment plans
- Upon satisfactory completion of the first three cases, reviewed by a member of the USAT, the referring clinician will receive a formal credentialing certificate, and your ‘scope of practice’ within GenesisCare practicing privileges will be updated to reflect eligibility to refer and treat linac prostate SABR at GenesisCare
- The GenesisCare website will be updated to note that you are now able to offer linac based prostate SABR treatment within the GenesisCare network.
- Peer review will continue for all linac prostate SABR cases as standard
Similarly, for MRIdian MR linac treatments a clinician can opt to enter the credentialing programme involving a presentation, contouring exercises and the first three cases to be mentored and peer reviewed.
All MRIdian MR linac cases are subject to ongoing peer review – a best practice to ensure safety and governance for all cases.
If you would like more information about how to access these treatments for your patients, please contact urologysat@genesiscare.co.uk
The Urology SABR Advisory Team (USAT)
The provision of 5# SABR for prostate cancer patients across the GenesisCare network is led and governed by the Urology SABR Advisory Team (USAT) to ensure clinical excellence and patient safety. The team consists of:
- Dr Philip Camilleri, Clinical Oncologist and Urology Clinical Director
- Dr Nicola Dallas, Clinical Oncologist, Specialist Advisor and MRI Linear Accelerator (MRL) Specialist
- Dr Prantik Das, Clinical Oncologist, Specialist Advisor and MRI Linear Accelerator (MRL) Specialist
- Dr Carla Perna, Clinical Oncologist, Specialist Advisor and MRI Linear Accelerator (MRL) Specialist
- Dr Dan Ford, Clinical Oncologist, Specialist Advisor and MRI Linear Accelerator (MRL) Specialist
These clinical oncologists are all members of the Urology Clinical Reference Group. They review all referrals of primary urological SABR treatments to assess patient eligibility for 5# SABR either on a standard linac or the MRIdian MR linac.
Referral process and clinical review
The USAT will review the suitability of all primary urological SABR treatments including prostate, on either a conventional linac or on the MRIdian, and prostate reirradiation and renal primary lesions on the MRIdian. Urological cases of oligometastatic disease will also be reviewed by our SABR Advisory team.
This team will review any submitted cases on Tuesdays and Fridays through an asynchronous eMDT. This ensures a fast turnaround and a prompt outcome for patients and their referring clinicians.
The urology MR linac/SABR advisory team referral form needs to be completed and submitted to the meeting coordinator along with the minimum data set which must include the following:
- Clinic letter
- Whole body and organ specific imaging with reports available for review in Carestream
- Histology (mandatory for prostate only)
This systematic review will ensure careful and appropriate case selection and maintain excellence in terms of clinical governance of the urology SABR service. Each review will generate an outcome report which in turn supports you and your patients to secure insurer preauthorisation for these state-of-the-art radiotherapy treatments.
The USAT is here to assist you and are always happy to advise you regarding this non-surgical treatment for prostate cancer.
For those yet to gain practicing privileges with GenesisCare, we are always looking to collaborate with talented consultants. We can help you build your private practice with medical secretary support, practice management systems, website and marketing support, free consultation rooms and over 300 education and engagement events per year. Our state-of-the-art IT solutions mean that tasks such as CT contouring, planning and plan approval can all be completed remotely.
Contact your local Referrer Engagement Manager (REM) to get started with the easy process.
References:
- Dearnaley D, Syndikus I, Mossop H, et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol 2016; 17: 1047–60
- Lee WR, Dignam JJ, Amin MB, et al. Randomized phase 3 noninferiority study comparing two radiotherapy fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol 2016; 34: 2325–32.
- Catton CN, Lukka H, Gu CS, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol 2017; 35: 1884–90.
- Mottet N, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol 2017; 71: 618–29.
- Sanda MG, Cadeddu JA, Kirkby E, et al. Clinically localized prostate cancer: AUA/ASTRO/SUO Guideline. Part 2: recommended approaches and details of specific care options. J Urol 2018; 199: 990–97
- Brand D, Tree A, Ostler P, van der Voet H, Loblaw A, Chu W et al. Intensity-modulated fractionated radiotherapy versus stereotactic body radiotherapy for prostate cancer (PACE-B): acute toxicity findings from an international, randomised, open-label, phase 3, non-inferiority trial. The Lancet Oncology. 2019;20(11):1531-1543.
- Kishan A. et al., Magnetic Resonance Imaging-Guided vs Computed Tomography-Guided Stereotactic Body Radiotherapy for Prostate Cancer: The MIRAGE Randomized Clinical Trial. JAMA Oncol. 12 Jan 2023. doi: 10.1001/jamaoncol.2022.6558.
SABR reirradiation
Reirradiation using stereotactic ablative radiotherapy (SABR)
As cancer prognosis continues to improve and the evidence to support treatment of oligometastatic disease increases, more patients have now received multiple courses of radiotherapy , either stereotactic ablative radiotherapy (SABR) or conventionally fractionated, and the scenario of reirradiation has become more frequent.
Reirradiation is made possible by technical advances, such as MR-guided radiotherapy, a SABR technique that enables high-dose radiation to limited volumes, with millimetric accuracy to help avoid critical healthy tissues. SABR is one of the most important advanced radiotherapy techniques currently available for treating tumours. The aim is to achieve local control or complete ablation in just a few short treatments, with longer disease-free intervals. Reirradiation is an important treatment choice for patients living with disease and having a good performance status, offering the potential for an improved quality of life. It also offers the possibility of staying off drug treatments, or reducing the frequency of drug switching, which is especially important if there are limited treatment options.
At GenesisCare, we have established our expertise in reirradiation using SABR through a careful multidisciplinary approach. We use rigorous physics and dosimetry to ensure the radiobiological aspects of treatment are accounted for. Our service is provided on the MRIdian MR linac (or the Varian Edge), which allows the volume of healthy tissue reirradiated to be kept to a minimum with daily adaptation and real-time visualisation.
For patients with pacemakers or with tumours in sites who would not be suitable for treatment on the MRIdian, reirradiation can also be delivered using the latest generation Varian Edge radiosurgery system at our Centre for Radiotherapy at Cromwell Hospital in London. The advanced capabilities of this linac integrate high precision with integrated machine intelligence. With automated gating and accuracy checks performed every hundredth of a second (10ms), this allows for safe and accurate reirradiation of tumours, whilst remaining minimally invasive. The Cromwell has on site cardiac technical support for patients with pacemakers.
Refer a patient
To refer a patient for reirradiation using stereotactic ablative radiotherapy at GenesisCare, complete our referral form or make an enquiry.
MR-guided SABR makes reirradiation a reality
The MRIdian technology combines magnetic resonance imaging (MRI) techniques with extremely precise radiotherapy beams, enabling the clinician to visualise the exact position and shape of a patient’s tumour in real time while their treatment is being delivered.
We were the first to introduce MRIdian technology to the UK at our centres in Oxford and Cromwell Hospital in London.
We're committed to growing the body of evidence through our team of expert SABR clinicians and our global collaborations as we continue to explore the possibilities of this technology and its role in modern cancer treatment.
Our expansion of this service for reirradiation of locally recurrent cancers reflects the growing demand from patients and clinicians and is based on numerous successful outcomes to date.
Reirradiation – treatment overview
SABR is one of the most important advanced radiotherapy techniques currently available. Using highly accurate targeting beams of high-dose radiation it can be administered with sub-millimetre accuracy through a minimal number of fractions. This approach retains the potential for effective ablation to achieve local control whilst minimising the risk of toxicity and damage to nearby organs at risk (OAR).
As the number of patients previously exposed to radiation treatment grows, demand is set to increase for new treatment options. With the advent of SABR, reirradiation is now an option in certain scenarios where in the past, toxicity concerns would have precluded further radiotherapy. In particular, the accuracy of SABR when delivered with MRI guidance opens up opportunities to present patients with an effective treatment option where previously no other options would have been available.
MR-guided protects organs at risk
At GenesisCare, we use MRIdian, an MR linac, to deliver reirradiation using SABR. Magnetic resonance image-guided radiotherapy (MRgRT) represents a step change in advanced radiotherapy, introducing new possibilities for reirradiation. MRIdian combines a high-definition MR scanner with a linear accelerator to provide maximum accuracy in the delivery of SABR.
Taking steps to reduce the risks of side effects and toxicity is crucial, particularly when treating areas that have already been exposed to doses of radiation. With MRIdian, the target area is visualised in real-time whilst treatment is administered. The treatment programme is adaptive, reacting to the slightest changes in order to limit exposure to radiation outside the treatment zone.
Find out more about MRIdian here
Reirradiation service at GenesisCare
Reirradiation SABR requires individualised planning to maximise the ablative dose whilst minimising toxicity. We use standardised dose and fractionation schedules based on tumour size and evidence-based best practices. Our dosimetrists and medical physicists calculate high-quality contouring plans, taking into account the normal tolerance of healthy tissue, organs at risk and inter-fractional adjustments. We use a sophisticated AI planning system to calculate target volumes, and all plans are peer-reviewed in multidisciplinary teams (MDTs).
The SABR Reference Group oversees all aspects of governance and service quality.
Who can I refer?
Reirradiation using SABR should be evaluated as a treatment option where the objective is to achieve local control. For some patients, SABR may be considered an alternative to chemotherapy or surgery, or it may be an option if surgery is either not possible or not desirable.
We are currently treating these tumour sites:
- Pelvic area – prostate
- Pelvic area – non-prostate
- recurrent gynaecological cancer
- recurrent anorectal cancer
- recurrent bladder / renal cancer
- Spine
- Lung
- Liver
- Pancreas
Patients who meet the following criteria can be referred to GenesisCare for reirradiation:
- Patient has received a previous course of radiotherapy to the proposed site with no significant long-lasting toxicity
- Ineligible for surgery with curative intent or surgery with curative intent is declined by the patient
- More than six months since the initial radiation treatment
- World Health Organisation (WHO) performance status ≤ 2
- Life expectancy of more than six months
Previous reirradiation to the proposed site or concurrent targeted therapies or systemic therapies generally preclude treatment.
For more information, including clinical data and case studies, please refer to our SABR evidence summaries.
Patient experience
From the patient’s perspective, reirradiation offers significant benefits in comparison with other treatment options:
- It’s non-invasive
- Typically delivered in one to five fractions
- Fast recovery time
- Greater convenience for patients
- Treatment is delivered as an outpatient procedure
- Treatment time is short – sometimes less than 30 minutes
- In some cases, there may be the potential for SABR to be delivered concurrently with other treatments, as an adjuvant or used as a bridge to other treatments
Reirradiation makes a difference for many patients, with the potential to offer an increase in life expectancy and an improvement in quality of life, including a reduction in pain.
At GenesisCare, we’re committed to offering patients the most appropriate treatment for their condition, with decisions made in conjunction with the patient and their wishes. We aim to utilise the most advanced techniques available to medical science today, offering patients the best possible outcomes.
Refer a patient
To refer a patient for reirradiation using stereotactic ablative radiotherapy at GenesisCare, complete our referral form or make an enquiry.
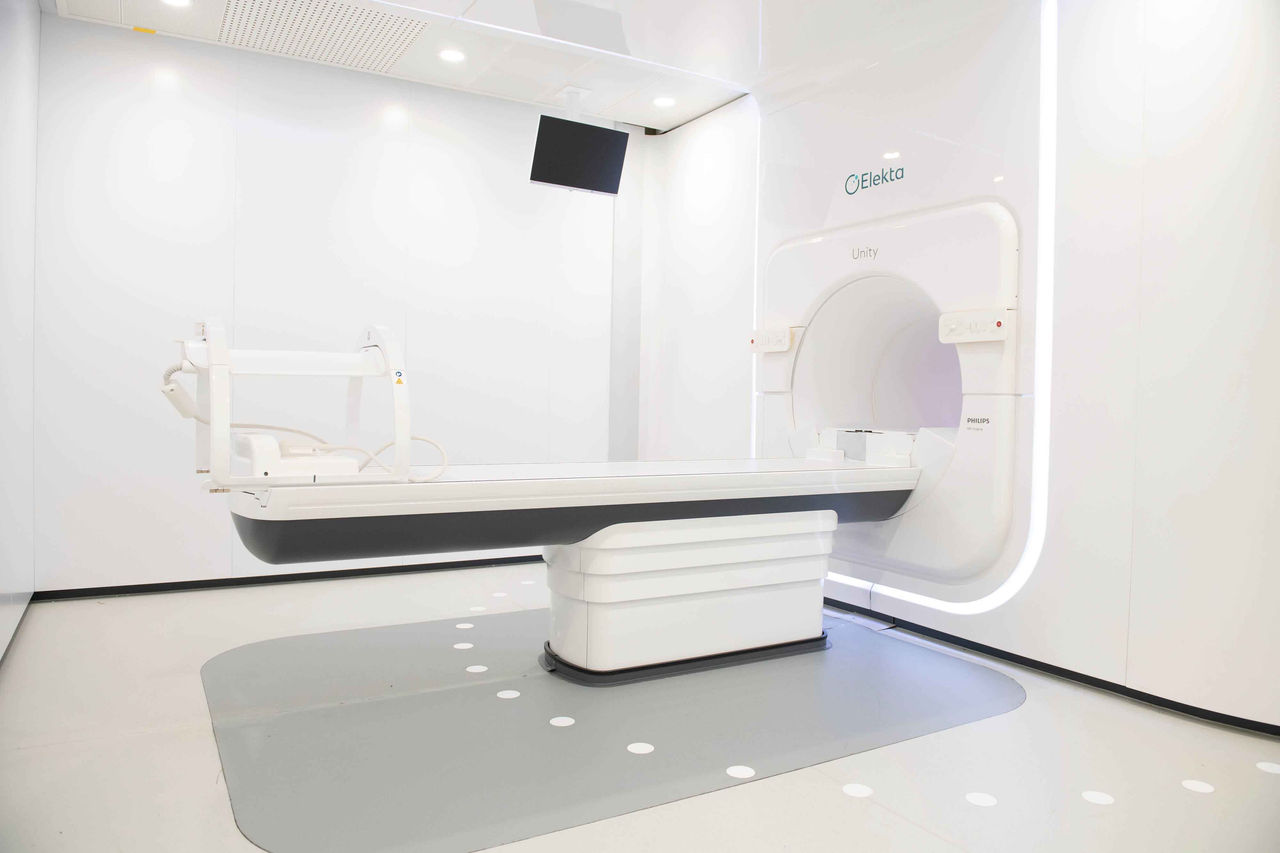
MRIdian MR-guided radiotherapy
Magnetic resonance image-guided radiotherapy, or MRIgRT, is the most exciting development in advanced radiotherapy for years. By combining a linear accelerator with a high definition MR scanner, MRIdian allows clinicians to see as they treat – adjusting the beam to allow for movements and anatomical changes. It provides a step-change in accurate stereotactic ablative radiotherapy (SABR), bringing a new level of control and confidence with tumour targeting and sparing of healthy tissue. MRIdian makes truly adaptive SABR possible for the first time.
With 10,000 patients treated to date worldwide, MRIdian has been widely used for gastrointestinal, lung and prostate cancer, and is increasingly recognised as a possible platform for partial breast treatments and safer re-irradiation. The possibility of incorporating on-table functional MRI into treatment pathways is just around the corner.
We’re proud to be the first independent healthcare provider in the UK to introduce the MRIdian at our centres in Oxford and Cromwell Hospital in London and can be accessed by patients throughout the UK. This is the foundation for an emerging MRIgRT network that will put us at the forefront of exploring the possibilities of this innovative approach. We are excited to be partnering with the University of Oxford and a team of expert SABR clinicians who will work with us to build a UK body of evidence that will impact the lives of many patients.
MRIdian, a new paradigm in radiation oncology
Real-time on-table adaptive radiotherapy brings a new level of confidence to SABR delivery
MRI-guided radiotherapy improves on conventional image-guided radiotherapy (IGRT) by providing improved soft tissue definition without additional radiation exposure through high-quality MR images.
By capturing MR images of the target many times a second, the MRIdian provides real-time moving images that capture the anatomical positional changes that occur naturally within the body. Uniquely, the MRIdian allows tracking of the target position in real time, coupled with a gating facility that prevents beam delivery when the target is outside the treatment boundaries. This level of accuracy means that uncertainty is reduced, eliminating the need for large margins around the target.
The MRIdian has opened up new avenues of treatment, particularly tumours that move and are within close proximity of organs at risk (OAR) such as tumours of the liver and pancreas, where high doses are achievable while keeping the doses to OARs within tolerance.
MRIdian MRlinac explained
How it works
The MRIdian allows clinicians to provide true on-line adaptive radiotherapy. The treatment planning system is clear and intuitive making daily re-contouring of tumour and normal tissues straightforward. The updated plans are rapidly re- optimised providing a personalised radiation treatment on every patient, every fraction.
- The MRIdian uses a patented split-magnet MR design, so that the radiation beam is not distorted by the magnetic field and is at the optimum distance for exceptionally sharp, high dose-rate SABR.
- The MR set-up captures multiple high-definition, high-quality images many times a second, seeing even the tiniest movement of the tumour and surrounding organs.
- The MRIdian performs real-time on-table plan adjustments to reflect both inter- and intra-fraction anatomical changes or movements.
- The gating target and gating boundary are visualised on the display. When the gating target moves outside the pre-specified gating boundary, the radiation beam automatically stops until the target returns back inside the gating boundary.
- Some patients benefit from being able to view this while inside the MRIdian – enabling them to take an active role in gated delivery with adjustments to their breathing.
- Coupled with the unique target tracking and gating system is a precise stereotactic radiation beam, giving the clinician greater control and confidence in delivering maximal dose to the tumour target while preserving critical structures.
The evidence base suggests that the MRIdian makes it possible to achieve ablative doses without reaching prohibitive levels of toxicity, particularly in the abdomen. This offers the prospect of improving clinical outcomes for previously hard-to-treat cancers such as locally advanced or recurrent pancreatic cancer and inoperable liver tumours.
Building a UK body of evidence
Through our academic collaboration with the University of Oxford, we will build on the body of evidence for MRIdian with an ongoing programme of clinical evaluation in the UK.
Our strategy for the introduction of MRIgRT is to align with international best practice based on evidence-based protocols. There is an increasing international evidence base for MR linac and notably the MRIdian system. Here are some of the pivotal studies that have informed our approach.
Prostate
A Phase III randomised clinical trial from the University of California has highlighted the benefits of using MRI guidance over CT guidance when treating patients with prostate cancer receiving 5# SABR at the same radiation dose. The MIRAGE trial showed that patients treated using MRI-guided SABR from MRIdian experienced significant reductions in the incidence rates of acute grade ≥2 genitourinary toxicity (43.4% vs. 24.4%, p = 0.01) and gastrointestinal toxicity (10.5% vs. 0.0%, p = 0.003) when compared to patients receiving CT-guided prostate SABR.1 This evidence helps address concerns surrounding early and late toxicity when using ultra hypofractionation.
The MRIdian makes it possible to deliver SABR with daily, real-time adaptive planning. This increases accuracy and reduces planning treatment margins, resulting in the reduced toxicity results shown in this study.
The MIRAGE trial demonstrates that, by using MRI-guided SABR, patients with prostate cancer experience fewer side effects and, therefore, have an improved quality of life compared to patients receiving CT-guided SABR.
Read more about MRIdian for prostate cancer.
5# SABR with simultaneous integrated boost (SIB)
We’re now offering SIB with MRI-guided 5# SABR for prostate cancer at our London and Oxford centres. This treatment option enables dose escalation to the dominant intraprostatic legion (DIL), the region of the prostate at highest risk of recurrence, whilst maintaining a favourable safety profile.
This approach is supported by the recent Phase III FLAME trial (Focal Lesion Ablative Microboost in Prostate cancer). In this study, patients who received a simultaneous integrated boost plus standard radiotherapy regimen showed improved outcomes compared to those who received the standard radiotherapy regimen alone.2,3 There were no significant differences in toxicity or quality of life between treatment regimens.4
The subsequent Hypo-FLAME trial demonstrated acceptable safety of SIB delivered with hypofractionated SABR radiotherapy.5
Find out more about simultaneous integrated boost.
Pancreas
A retrospective study of 44 patients with inoperable pancreatic cancer evaluated the overall survival benefit of higher dose, adaptive MR-guided SABR versus standard, low-dose SABR.6 High-dose MRIgRT patients (n = 24) had a statistically significant improvement in 2-year overall survival (OS) (49% vs 30%, P = 0.03) and trended towards significance for 2-year freedom from local failure (FFLF) (77% vs 57%, P = 0.15) compared to standard- dose patients (n = 20, 45%). This improved OS was achieved without any increased toxicity in the high-dose group. MRIgRT is now being tested in a prospective trial, which through our academic partners at the University of Oxford, GenesisCare will be supporting.
Read about our Rapid access to SABR for pancreatic cancer
Kidney
Treatment on the MRIdian MR Linac allows for a reduction in radiation dose to the surrounding normal tissues, such as the small bowel, spinal cord, large bowel and second kidney7, reducing the risk of possible toxicity which may otherwise be caused. It also offers a reduced risk of diminished renal function when compared to other interventions.
In a study of 95 patients with larger (T1b, >4 cm) renal cell carcinoma, where 77.6% were defined as inoperable by the referring urologist. 38 patients (40%) had a grade 1 to 2 toxicity and no grade 3 to 5 toxicities were reported. Cancer-specific survival, overall survival, and progression-free survival were 96.1%, 83.7%, and 81.0% at two years and 91.4%, 69.2%, 64.9% at four years, respectively.8
Read more about MRIdian for kidney cancer
Rectum
Phase II and III trials evaluating short-course radiotherapy as part of total neoadjuvant therapy for rectal cancer have shown encouraging results.9-11Delivered with MRI guidance, dose escalation offers an organ preservation strategy whilst maintaining a favourable toxicity profile.12
Research into using MRI-guided radiotherapy in rectal cancer is at an early stage. However, several recent studies have shown promising potential for this treatment modality:
- A study of 22 patients with rectal cancer delivering long-course chemoradiotherapy using MRI guidance demonstrated it to be a feasible treatment option, with 3 patients achieving complete clinical response and 4 patients a pathological major response.13
- A separate feasibility study of 5 patients with rectal cancer used an MR-linac to deliver weekly response-adaptive boost fractions in addition to conventional radiotherapy. 14Treatment was feasible and well-tolerated by all patients, with 4 reaching a clinical complete response. Quality of life and continence scores during follow-up were comparable to scores at baseline.
- A study of 43 patients receiving 25 Gy in 5 factions for rectal cancer explored how daily online adaptation of the clinical target volume using MRI guidance can reduce the margins of the MRIdian’s planning target volume (PTV). Margins were reduced for 18 patients and the median in-room time per fraction was recorded at 48 minutes, demonstrating the feasibility of this strategy
- MR-guided SCRT (standard dose) on 1.5T MR Linac with n=43 patients: 100% compliance rate; median treatment time 48 minutes, CTV margin reduction in 40% (Intwen et al Radiotherapy Oncol 2021)
- MR-guided SCRT (standard dose) n=15 patient. Planning study on 1.5T MR Linac. Intrafraction motion required 9mm margin with standard treatment time, could be reduced to 4mm if treatment within 15min (Eijkelenkamp Radiother Oncol 2021)
- MR- guided LCCRT (SIB to 55Gy in 25#) n=35 patient. Prospective feasibility study on MRIdian. G3+ toxicity profile 20% (Chiloiro et al CTRO 2021)
- MR guided LCCRT (n=1) Two phase plan 50/45Gy in 25# plus three weekly MR guided 3Gy boost fractions) 1.5 T MR-Linac, the authors observed high response rates, excellent organ function and limited side effects (Gani et al Radiother Oncol 2021)
- MR guided dose adapted LCCRT (Thunder 2) with decision about dose escalation (60Gy in 25# SIB) based on radiomics features on MRIdian
- MR guided SIB (9Gy in 5 fractions) on 1.5T MR Linac followed by LCCRT (45Gy in 25#) on conventional linac n=12 patients PTV margin reduced to 3-5mm even without tracking/gating functionality (Ingle et al, Clin Oncology 2023)
- MR guided SIB LCCRT feasibility study with 36Gy in 20# on a conventional linac followed by two phase boost 9Gy/5# to 16.5Gy/5# to primary on 1.5T MRL (ESTRO 2023 RMH)
- PreRADAR – phase I feasibility dose escalation study of standard SCRT followed by MR guided sequential boost (2x5Gy to 5x5Gy) on 1.5T MRL (Verweij et al BMJ Open 2023).
At GenesisCare, we’re committed to growing the evidence base for MRI-guided radiotherapy and will be publishing results from our own research later this year.
Our MRIdian case studies
MR linac is at the cutting edge of what is currently possible in radiotherapy technology. The type of patients we treat using this modality include many that could not otherwise be safely treated to a similar dose on other platforms. Read some of our case studies.
Cancers treated
The evidence base for SABR is growing and there is currently a range of clinical scenarios into which it can be integrated. At GenesisCare, we offer SABR in the following situations:
- Localised prostate cancer, where hypofractionated treatment offers an alternative to conventional therapy
- Early stage lung cancer
- Gastric lymphoma (MALT and diffuse large B cell lymphoma [DLBCL]) after chemotherapy and residual visible abnormality in patients with bulky Hodgkin’s and Non-Hodgkin’s lymphoma after initial systemic anti-cancer therapy
- Hepatobiliary cancer, including primary liver cancers and inoperable/recurrent pancreatic cancer
- Cervical cancer
- Pelvic re-irradiation
- Selected cases of oligometastatic and oligo- progressive cancers
- Renal cell carcinoma where surgery is not possible or not preferrable, and other treatments are not advisable (e.g. for larger tumours where risk of damage to surrounding organs is greater, or central kidney tumours where surgery is not possible and SABR would preserve greater renal function)
We have defined SABR referral criteria based on the experience of our SABR clinical expert team, published clinical studies and international best practice. All SABR referrals will be assessed for a possible benefit of treatment on MRIdian. For example:
- Tumours where there is significant movement, e.g. in the upper abdomen and lung
- Tumours that are in close proximity to organs at risk (OARs), e.g. in the prostate, upper abdomen
and central lung
For other tumour sites, please refer to our referral criteria via the MOSAIQ tool available on our referral portal.
Delivering clinical excellence
We are committed to an environment of clinical excellence, objectivity and accountability. Our processes are adapted from international best practice and our team has undertaken training with centres including Amsterdam UMC, Miami Cancer Institute, Henry Ford Cancer Centre (University of Wisconsin) and Heidelberg University Centre.
The introduction of MRIdian has been overseen by Specialist Reference Groups in both urological cancers and SABR. The groups are comprised of clinicians who have undergone extensive MRIdian training and have a vast cumulative experience of delivering highly technical radiotherapy. The groups work closely together in a robust forum to guide all aspects of clinical governance, patient selection, peer review, education and credentialing. The groups also oversee research activity, developing joint protocols with our academic partners. GenesisCare will treat more patients with MRIgRT than any other institute globally, and will therefore be at the vanguard of translating research into everyday precision radiation medicine.
Find out more about clinical leads and governance at GenesisCare
References
- Kishan A. et al., Magnetic Resonance Imaging-Guided vs Computed Tomography-Guided Stereotactic Body Radiotherapy for Prostate Cancer: The MIRAGE Randomized Clinical Trial. JAMA Oncol. 12 Jan 2023. doi: 10.1001/jamaoncol.2022.6558.
- Kerkmeijer LGW, Groen VH, Pos FJ, Haustermans K, Monninkhof EM, Smeenk RJ, Kunze-Busch M, de Boer JCJ, van der Voort van Zijp J, van Vulpen M, Draulans C, van den Bergh L, Isebaert S, van der Heide UA. Focal Boost to the Intraprostatic Tumor in External Beam Radiotherapy for Patients With Localized Prostate Cancer: Results From the FLAME Randomized Phase III Trial. J Clin Oncol. 2021 Mar 1;39(7):787-796. doi: 10.1200/JCO.20.02873. Epub 2021 Jan 20. PMID: 33471548.
- Groen VH, Haustermans K, Pos FJ, Draulans C, Isebaert S, Monninkhof EM, et al. Patterns of Failure Following External Beam Radiotherapy With or Without an Additional Focal Boost in the Randomized Controlled FLAME Trial for Localized Prostate Cancer. Eur Urol [Internet]. 2022;82(3):252–7. Available from: https://www.sciencedirect.com/science/article/pii/S0302283821022235
- Monninkhof EM, van Loon JWL, van Vulpen M, Kerkmeijer LGW, Pos FJ, Haustermans K, et al. Standard whole prostate gland radiotherapy with and without lesion boost in prostate cancer: Toxicity in the FLAME randomized controlled trial. Radiotherapy and Oncology. 2018 Apr 1;127(1):74–80.
- Draulans C, van der Heide UA, Haustermans K, Pos FJ, van der Voort van Zyp J, De Boer H, et al. Primary endpoint analysis of the multicentre phase II hypo-FLAME trial for intermediate and high risk prostate cancer. Radiotherapy and Oncology [Internet]. 2020 Jun 1;147:92–8. Available from: https://doi.org/10.1016/j.radonc.2020.03.015
- Rudra, S. et al. Using adaptive magnetic resonance image-guided radiation therapy for treatment of inoperable pancreatic cancer. Cancer Med. 8, 2123–2132 (2019).
- Siva S, Ellis RJ, Ponsky L, Teh BS, Mahadevan A, Muacevic A, Staehler M, Onishi H, Wersall P, Nomiya T, Lo SS. Consensus statement from the International Radiosurgery Oncology Consortium for Kidney for primary renal cell carcinoma. Future Oncol. 2016 Mar;12(5):637-45
- Siva S, Correa R, Warner A, Staehler M, Ellis R, Ponsky L et al. Stereotactic Ablative Radiotherapy for ≥T1b Primary Renal Cell Carcinoma: A Report From the International Radiosurgery Oncology Consortium for Kidney (IROCK). Int. J. Radiat. Oncol. Biol. Phys. 2020;108(4):941-949.
- Erlandsson, J. et al. Radiotherapy regimens for rectal cancer: long-term outcomes and health-related quality of life in the Stockholm III trial. BJS Open 5, (2021).
- Jin J et al. Multicenter, Randomized, Phase III Trialof Short-Term Radiotherapy Plus ChemotherapyVersus Long-Term Chemoradiotherapy in LocallyAdvanced Rectal Cancer (STELLAR). J Clin Oncol 40, 1681–1692 (1011).
- Bahadoer, R. R. et al. Short-course radiotherapy followed by chemotherapy before total mesorectal excision (TME) versus preoperative chemoradiotherapy, TME, and optional adjuvant chemotherapy in locally advanced rectal cancer (RAPIDO): a randomised, open-label, phase 3 trial. Lancet Oncol 22, 29–42 (2021).
- Boldrini, L., Intven, M., Bassetti, M., Valentini, V. & Gani, C. MR-Guided Radiotherapy for Rectal Cancer: Current Perspective on Organ Preservation. Front Oncol 11, (2021).
- Chiloiro, G. et al. MR-guided radiotherapy in rectal cancer: First clinical experience of an innovative technology. Clin Transl Radiat Oncol 18, 80–86 (2019).
- Boeke, S. et al. Online MR guided dose escalated radiotherapy for organ preservation in distal rectal cancer. Clin Transl Radiat Oncol 37, 153–156 (2022).
- Intven, M. P. W. et al. Online adaptive MR-guided radiotherapy for rectal cancer; feasibility of the workflow on a 1.5T MR-linac: clinical implementation and initial experience. Radiotherapy and Oncology 154, 172–178 (2021).
Image-guided radiotherapy (IGRT)
We use image-guided radiotherapy (IGRT) based on 3D cone beam verification immediately prior to treatment for accurate set-up. These are compared and matched with simulated X-rays taken directly from 3D CT data used for treatment planning. Our radiographers have extensive experience of CBCT interpretation and use of specialist positioning equipment to ensure submillimetre accuracy in set-up and minimal delay.
We are one of the first companies to introduce MRI-guided radiotherapy (MRIgRT) for cancer treatments with the MRIdian. This is an exciting step forward in the real-time planning and adaptive treatment of tumours and is available for patients across the UK. MRIdian is available for prostate, lung, liver and pancreatic cancer and is an important part of these treatment pathways.
Surface-guided radiotherapy (SGRT)
We use the latest surface-guided radiotherapy (SGRT) techniques in combination with VMAT or SABR to enable 3D and 4D set-up and superior motion management during radiotherapy.
The accuracy of radiotherapy is enhanced when combined with SGRT – a sophisticated stereovision technology that monitors thousands of points on the patient’s skin during set-up and treatment, unlike other commonly used systems that rely on CT imaging – further increasing radiation exposure.
This powerful system reduces positioning errors to improve the overall accuracy, as well as the speed and comfort of treatment.
Examples are:
- Gating and beam hold facilitating 4D IMRT for moving lesions
- SABR and SRS couch and patient monitoring for accurate set-ups
- Intrafraction variation minimisation by capturing surface data in real time
- Surface data captured at each attendance to manage interfraction variation and dosimetric changes
These advances provide important benefits to our patients including the option of open-face treatment shells and tattoo-free treatments.
Stereotactic radiosurgery (SRS)
Stereotactic radiosurgery
Stereotactic radiosurgery (SRS) is often described as brain area-sparing surgery and has revolutionised the treatment of brain metastases.
It uses high-quality imaging to deliver precisely-focused radiation beams at high doses in a single or few short treatments. SRS is an advanced approach that permits the maximum conformal dose delivery within the targets while minimising dose to the surrounding unaffected brain. High dose treatment with sufficient dose intensity helps to ablate the tumour cells and aims to increase the local control of the disease whilst preserving important brain function.
The latest evidence demonstrates that treatment with SRS alone results in less decline in cognitive deterioration and better quality of life than SRS with whole brain radiotherapy. Vitally, this is achieved with no significant difference in overall survival.1 SRS is now replacing conventional whole brain radiotherapy as the procedure of choice for some primary tumours and solitary or multiple brain metastases.
SRS is central to our neuro-oncology service at GenesisCare as we seek to offer our patients the option of achieving an optimum life outcome for them and their families. We have invested heavily in SRS technology and expertise, as part of an end-to-end pathway that includes early detection, treatment, close monitoring and follow-up. In the management of brain metastases, an approach that focuses on patient quality of life and functional outcome is becoming the norm.
GenesisCare is a specialist provider of neuro-oncology and a centre of excellence for stereotactic radiosurgery for private patients across the UK. We have invested in an end-to-end solution that involves brain-sparing technologies and innovative protocols, delivered through a team of leading clinicians at our centres in London and Oxford.
Refer to us
If you would like further information about our neuro-oncology service, please contact your Referrer Engagement Manager: MDM@genesiscare.co.uk
Stereotactic radiosurgery – a gold standard treatment
Brain metastases affect up to 30% of all cancer patients and are the most common sequelae of cancer. Lung cancer, breast cancer, kidney cancer and melanoma are the most common primary tumours that metastasise to the brain.
Stereotactic radiosurgery (SRS) has become increasingly recognised worldwide as the gold standard radiotherapy option by professional societies, based on a growing evidence base for the use of SRS alone for a patient presenting with one to four brain metastases.2
SRS is routinely used in re-irradiation after brain radiotherapy or previous SRS. SRS is shown to have the same overall survival in two to four metastases as five to ten.3
Use of stereotactic radiosurgery (SRS) is no longer limited to patients with three or fewer lesions, because data suggest that total disease burden, rather than number of lesions, is predictive of survival benefits associated with the technique.
- SRS is increasingly becoming an integral part of management of patients with controlled, low‑volume brain metastases. National Comprehensive Cancer Network (NCCN) Guidelines, 2014.
- Don’t routinely add adjuvant whole brain radiation therapy to stereotactic radiosurgery for limited brain metastases. American Society for Radiation Oncology (ASTRO), Choosing Wisely Campaign, 2014.
- It is the recommendation by ISRS that patients with one to four brain metastases with reasonable performance status and prognosis be treated with SRS alone, with whole brain radiotherapy (WBRT) reserved as one of many salvage therapeutic options. International Stereotactic Radiosurgery Society consensus.
Who we treat
We have experience of managing a wide range of brain and CNS tumours. Patients can be referred to our Neuro-oncology Advisory Team in Oxford or London for review and assessment. Referring clinicians are involved in decisions and are assured a fast response within five days.
Brain metastases:
- Brain metastases with a combined total volume of 20cc
- Life expectancy > 6 months
- Histological diagnosis of cancer
- Potentially controllable extracranial disease
- Good performance status (PS-01)
Re-treatment with SRS is possible:
- After 3 months for a new metastases
- After 6 months for a previously treated metastases
Contraindications for SRS:
- Large volume brain metastases (>20cc)
SRS is also indicated for:
- Meningiomas, acoustic neuromas and primary pituitary tumours
- Other non-cancer conditions including arteriovenous malformations and complex trigeminal neuralgia
Neuro-oncology at GenesisCare
Our ambition has been to design a neuro-oncology service that treats the whole patient, not just the tumour. The incidence of brain metastasis is increasing, due to better imaging surveillance and improved systemic treatment of extracranial disease. Conventional radiotherapy treatments such as whole brain radiotherapy have a higher risk of developing cognitive impairment and limited benefit in terms of survival. Advanced techniques such as stereotactic radiosurgery (SRS) offer improved quality of treatment, delivering higher radiation dose to the tumour whilst reducing the impact on the normal brain and thereby decreasing toxicities.
Tumours of the brain and central nervous system are a particular specialty for us and we are a specialist independent provider of neuro-oncology in the UK and a centre of excellence for stereotactic radiosurgery (SRS).
SRS at Oxford
At our Oxford centre SRS is delivered using Brainlab Exactrac, a fast and highly accurate visualisation and patient position monitoring system that is seamlessly integrated with a state-of-the-art Elekta Versa linear accelerator. It is installed alongside a specialist treatment planning system. It employs two low-dose X-ray images, which are captured from two different angles. They are compared and matched with simulated X-rays taken directly from 3D CT data used for treatment planning. A robotic treatment table adjusts for patient position with sub-millimetre movements – maintaining the accuracy of the procedure and ensuring the radiation treatment dose is delivered as prescribed.
Due to the large number of patients treated, our specialist planning and clinical teams are experts in precise dose planning and efficient patient set-up. This ensures smooth and professional patient care, with no waiting around.
SRS at London
At our GenesisCare radiotherapy centre at Cromwell Hospital in London, we have two advanced systems in SRS delivery: Elekta's Leksell Gamma Knife Icon and Varian Edge with HyperArc. Gamma Knife Icon has the flexibility to enable personalised, high-dose treatment plans across a wide range of diagnoses. It builds on the established precision of Gamma Knife and offers single or fractionated frame-based or frameless treatments with precision and accuracy.
The system is integrated with all components, including a 3D cone-beam CT, built on the same rigid structure and calibrated to one another to improve precision. During treatment, real-time motion management monitors the patient with a resolution of 0.15mm. If the patient moves outside a pre-set limit, beam delivery automatically turns off. These features lead to the increase in precision and accuracy. Studies have demonstrated that Gamma Knife Icon can deliver a 2-4x lower dose to the normal brain, when compared with earlier systems.4
Varian Edge with HyperArc enables clinicians to efficiently plan and deliver high-quality SRS for simple or complex cases with multiple metastases. HyperArc allows treatment planners to create high-quality plans efficiently and without the need for a physical dry-run, thanks to treatment visualisation tools. A one-click workflow automates most of the planning and delivery process so teams can focus on other objectives, such as target definition and dose, and SRS can be delivered within 20 minutes. Planning algorithms allows single isocentre planning for multiple metastasis cases, whilst minimising bridging doses in normal brain. Planners also have the flexibility to use multiple isocentres if more appropriate for the patient's treatment.
In a study comparing HyperArc VMAT planning and conventional VMAT planner, HyperArc achieved an improved plan quality with higher conformity index (CI), sharper dose falloff and better sparing of normal brain tissue.5 Another study showed HyperArc plans were comparable with CyberKnife plans.6
Peer-to-peer team-working in a secure environment
Our multi-disciplinary structure is pivotal to our neuro-oncology service. Bringing together the expertise of neuroradiologist, neuroimaging scientists, neuro-oncologists and neurosurgeons alongside the planning and delivery team which includes physicists and radiographers, we can tailor care to optimise outcomes for each and every patient.
To facilitate this in an independent setting, we have established a bespoke neuro-oncology eMDT (multidisciplinary team) platform which allows peer-to-peer collaboration, accessible at any time. The eMDT enhances clinical processes, using real-time data tracking and video to enable the neuro-oncology team and wider clinical teams to discuss patient care plans while protecting the flow of sensitive data. It will also audit decision-making and outcomes data within a best practice framework. We are the first independent provider to implement a fully integrated eMDT for neuro-oncology.
Our streamlined Rapid access SRS pathway allows appropriate patients to have SRS within just one week of the treatment decision.
Referring clinicians are invited to access our eMDTs by completing a Neurology Proforma.
A focus on clinical excellence for neuro-oncology
We are committed to creating an environment of clinical excellence for SRS and neuro-oncology services. Our protocols are adapted from international best practice and overseen by a core team of specialists, selected on the basis of their extensive experience and high-quality credentials. Their key role is to align the neuro-clinical service with evidence-based treatments such as SRS to ensure safe, consistent care across all of our centres. This robust forum guides all aspects of clinical governance, patient selection, peer review, education, research and assessment of new clinicians to the neuro-oncology team. In particular, the Clinical Reference Group form the backbone of our eMDT system – a multidisciplinary teamworking platform that connects our experts with a wider group of referring clinicians to discuss and review patient care.
The Neuro-oncology Multidisciplinary Team (MDT) reviews all referrals and assesses suitability for SRS.
Patient safety is at the heart of what we do
We are CQC inspected and have a comprehensive governance framework that ensures safe treatment protocols, evaluates consultant practising privileges and reviews cases outside standard treatment protocols.
Refer to us
If you would like further information about our neuro-oncology service, please contact your Referrer Engagement Manager: MDM@genesiscare.co.uk
References
- Brown et al., 2016; JAMA; 316 (4):401-409.
- Chao T. et al., 2018; Neurosurgery; 83 (3): 345–353.
- Yamamoto et al., 2014; Lancet Oncol; 15:387-95.
- Ma L et al., 2014; Int J CARS; 9:1079-1086
- Ohira S, et al., 2018; Rad Onc; 13(1)
- Kadoya N, et al., 2019; Med Dos; 44(4).394-400
Gamma Knife radiosurgery for benign conditions
A centre of excellence for stereotactic radiosurgery
GenesisCare is a global organisation and the UK’s leading independent provider of advanced and innovative radiotherapy and cancer therapies. We collaborate with health professionals who share our vision to ensure fast access to world-class cancer care and our strategy is to invest early in new, evidence-based modalities that hold potential for improved life outcomes.
Tumours of the brain and central nervous system, both benign and cancerous, are a particular area of expertise for us as a specialist independent provider of neuro-oncology in the UK and a centre of excellence for stereotactic radiosurgery (SRS). We have invested heavily in SRS technology and expertise as part of an end-to-end pathway that includes early detection, treatment, close monitoring and follow-up.
In 2020 we announced the launch of our SRS services at our GenesisCare Centre for Radiotherapy at Cromwell Hospital in London, which includes our state-of-the-art Gamma Knife Icon, and innovative protocols for benign brain conditions, delivered through a team of leading clinicians.
We have been at the forefront of introducing the routine use of DIBH for all left-sided breast cancer patients. We are the first independent oncology provider in the UK to formally implement DIBH for right-sided breast cancers, as standard.
Get in touch
We welcome referring neurosurgeons, neurologists, oncologists, and GPs to contact us to find out how we can help you and your patients.
We also welcome enquiries from any neurosurgeons, oncologists or related specialists who wish to partner with us by applying for practising privileges with GenesisCare.
We have created a world class end-to-end stereotactic radiosurgery pathway which includes fMRI and eMDT
- GenesisCare
Gamma Knife radiosurgery for benign conditions
Gamma Knife radiosurgery is a non-surgical radiation therapy and a well-established SRS technique that delivers large doses of highly targeted radiation to treat tumours and benign conditions including:
- Vestibular schwannoma
- Pituitary adenoma
- Meningioma
- Other skull base tumours
- Arteriovenous malformation
- Trigeminal neuralgia
- Some forms of epilepsy, Parkinson’s disease and tremors
SRS, often described as brain area-sparing surgery, is an advanced approach that uses high-quality imaging to deliver precisely-focused radiation beams at high doses in a single or few short treatments. This permits the maximum conformal dose delivery within the targets while minimising dose to the surrounding unaffected tissue preserving important brain function.
SRS is well tolerated by patients, making the list of exclusions minimal. Patients can be referred to our Neuro-oncology Advisory Team for review and assessment. Referring clinicians are involved in decisions and are assured a fast response with MDT recommendations. SRS may not be suitable if the treatment area includes important nerves which could be accidentally damaged or the volume of disease is more than 20cc.
A new level of precision
Our specialist radiotherapy centre at Cromwell Hospital offers the most advanced Leksell Gamma Knife Icon system for SRS, enabling a new level of accuracy in a single or fractionated dose. This next generation system has the flexibility to enable more personalised, high dose treatment plans across a range of patient types. It builds on the established precision of Gamma Knife and has the technology to enable single or fractionated frame-based or frameless treatments.
The Gamma Knife Icon is integrated with all components including a 3D cone-beam CT, built on the same rigid structure and calibrated with the Icon to improve precision. During treatment, real time motion management monitors the patient with a resolution of 0.15mm. If the patient moves outside a pre-set limit, beam delivery automatically turns off. Gamma Knife Icon can deliver a two to four times lower dose to the normal brain compared to earlier systems1.
We work with a team of neuro-oncologists, neurosurgeons, and neuro-radiologists, who have extensive experience of the Gamma Knife system and can optimise treatment for every patient.
1. Ma L et al., 2014; Int J CARS; 9:1079-1086
Unique diagnostic and planning capabilities
At GenesisCare we use advanced diagnostics and treatment planning techniques, such as fMRI and diffusion tractography, to personalise treatments and improve quality of life outcomes for benign brain tumours. fMRI and DTI are currently available in our Oxford centre. Patients can be referred from other centres as required. When required, we will help our patients with transport.
Multi-modal MR imaging guides cognitive and neurological outcomes
A non–invasive MRI technique, fMRI, is used to locate eloquent brain tissue related to speech, vision, movement, etc. It is used to guide neurosurgical planning and tailor surgery for intrinsic brain tumours to avoid neurological deficit and improve patient outcomes.
With the increasing neuro-oncological benefits of surgery for brain tumours, fMRI offers further potential applications to measure brain recovery processes to optimally stage and monitor treatments. In addition, patients are offered a complementary MRI technique called diffusion tensor imaging (DTI). This imaging scan provides assessment of tissue microstructure and we use tractography to provide a 3D visualisation of neural tracts and the brain’s connectivity. This provides additional information about the location of important pathways for speech, movement, vision and other behaviours important for patient quality of life, whereas conventional MRI techniques provide only anatomical information.
This service is not generally available at other providers.
Lower dose to healthy tissue
Gamma Knife radiosurgery is well established and offers high levels of patient acceptance. It is not painful and can be delivered in a single, outpatient session and completed in a few hours, allowing patients to quickly return to their normal life.
Side effects minimised by reduced radiation
The meticulous planning and treatment processes involved in SRS at GenesisCare, together with the reduction in radiation to the healthy brain help to minimise side effects. Not everyone experiences side effects but the patient’s risk may be affected by their general health, other treatments they may have had or the location of the lesion or tumour. Some patients may experience short-term side effects which usually disappear within a few weeks of treatment.
The treating clinician will discuss potential side effects in detail with the patient as part of informed consent. Around <10% of people notice long term side effects which are related to the treatment area within the brain. These may include:
- Hair loss from the area treated
- Hearing loss
- Loss of brain function
- Memory loss
- Pituitary gland damage
- Radiation damage to the normal brain
- Visual impairment
A focus on clinical excellence
We are committed to creating an environment of clinical excellence for SRS. Our protocols are adapted from international best practice and overseen by a core team of specialists, selected on the basis of their extensive experience and high-quality credentials. Their key role is to align the neuro-clinical service with evidence-based treatments such as SRS to ensure safe, consistent care across all of our centres.
This robust forum guides all aspects of clinical governance, patient selection, peer review, education, research and assessment of new clinicians to the neuro-oncology team. In particular, the Neuro-oncology Advisory Team form the backbone of our eMDT system – a multidisciplinary teamworking platform that connects our experts with a wider group of referring clinicians to discuss and review patient care.
Patient safety is at the heart of what we do. We have a comprehensive governance framework that ensures safe treatment protocols, evaluates consultant practising privileges and reviews cases outside standard treatment protocols.
These innovations are crucial to our approach – treat the whole patient, and not just the condition
- Mr David Peterson, Consultant Neurosurgeon
Peer-to-peer team-working in a secure environment
Our multidisciplinary structure is pivotal to our Gamma Knife radiosurgery service. Bringing together the expertise of neuro-radiologists and neuroimaging scientists, neurosurgeons and neuro-oncologists, we can tailor care to every patient and optimise outcomes for high-risk cases.
To facilitate this in an independent setting, we have established a bespoke eMDT platform which allows peer-to-peer collaboration, accessible at any time. The eMDT enhances clinical processes, using real-time data tracking and video to enable the neurology team and wider clinical teams to discuss patient care plans while protecting the flow of sensitive data. It will also audit decision-making and outcomes data within a best practice framework. We are the first independent provider to implement a fully integrated eMDT for neuro-oncology.
Get in touch
We invite referring neurosurgeons, neurologists, oncologists, and GPs to contact us to find out how we can help you and your patients.
Deep Inspiration Breath Hold (DIBH)
We have been at the forefront of introducing the routine use of DIBH for all left-sided breast cancer patients. We are the first independent oncology provider in the UK to formally implement DIBH for right-sided breast cancers, as standard.
Non-melanoma skin cancer (NMSC) is the most common cancer in the UK. Although it can arise in isolation, a significant group of patients have extensive skin field cancerisation (ESFC) where pre-cancerous lesions (actinic keratosis) pre-exist with cancerous lesions – basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). This carries a high risk of recurrence of pre-cancerous and cancerous lesions, requiring repeated treatment over many years. The ongoing risk of skin cancer and the physical and cosmetic impacts of disease and interventions can affect the patients’ quality of life.
The new standard of care
As the UK’s leading independent oncology specialist, we are dedicated to providing the latest evidence-based treatments from around the world, in a high quality, safe environment – without delay.
We have been at the forefront of introducing the routine use of Deep Inspiration Breath Hold (DIBH) for all left-sided breast cancer patients receiving radiotherapy (RT), based on strong evidence this reduces radiation dose to the heart and lung. DIBH is also routinely used for all patients treated with a VMAT technique, including delivering regional nodal irradiation (RNI) to the internal mammary chain (IMC), regardless of laterality.
We are the first independent oncology provider in the UK to formally implement DIBH for right-sided breast cancers, as standard, as it has been shown that it provides clinically significant reductions in potentially toxic right lung doses and mean liver dose.
Refer a patient
To refer a patient for breast cancer radiotherapy at GenesisCare, complete our referral form or make an enquiry.
Evidence-base for DIBH
DIBH is easily performed by the majority of patients and adds very little to treatment delivery times. The technique used at GenesisCare is an innovative response to tumour motion, developed using 3D surface monitoring and automated gating technologies to displace organs at risk (OAR) from the high-dose region of the tangential radiation field. Many groups have shown the benefits of DIBH, both in left, and more recently right-sided radiotherapy, particularly when the internal mammary chain (IMC) requires treatment.
Dosimetric studies show DIBH for right-sided breast cancers, with or without regional nodal irradiation (RNI), provides clinically significant reductions in potentially toxic right lung doses and mean liver dose. Although hepatic dose is not usually a concern, it is recognised that free breathing treatment delivery potentially underestimates the amount of radiation the liver would receive.
DIBH further improves safety margins, enabling more accurate treatment and minimising toxicity and dose to OARs in line with the ‘as low as reasonably practicable’ (ALARP) principle. The recent implementation of the hypofractionated, high dose-per-fraction schedule from the Fast Forward trial into routine clinical practice makes accurate planning target volume and OAR dose delivery more crucial than ever.
Radiotherapy expertise for DIBH
Radiotherapy is rapidly evolving, with new treatment modalities offering reduced acute and long-term side effects and significantly improved life outcomes for curative and palliative patients. GenesisCare is at the forefront of the introduction of innovative and advanced techniques. Our centres are equipped with the latest-generation linear accelerators and new modalities as standard.
Pre-treatment process
The patient will be trained by the radiographers on how to achieve and hold a stable breath hold prior to the CT planning scan. Two scans will be performed at the planning CT visit. The first (free breathing) scan helps position the patient on set using SGRT, with no need for permanent ink tattoos. The second (breath hold) scan is then taken and used for planning and treating the patient.
DIBH with state-of-the-art motion monitoring technologies
We use the latest surface-guided radiotherapy (SGRT) techniques – AlignRT – together with VMAT 3D and 4D set-up, and automated beam gating, which automatically stops treatment when the patient moves out of position with the aligned planning CT. This is combined with DIBH techniques to displace OARs from the high-dose field and stabilize the PTV. The technique is well tolerated by patients and improves comfort compared with spirometry.
Latest generation VMAT
VMAT is the next generation of intensity-modulated radiotherapy (IMRT), delivering a highly conformal radiotherapy dose by changing intensity as it moves around the tumour site. This reduces treatment time as well as the overall toxicity of treatment. VMAT is used to treat many cancer types, including breast, where we are pioneering the use of Partial Breast Irradiation.
We provide VMAT to all suitable patients and use simple automation and Physics QA automation (Mobius) to create highly conformal plans for dosimetry.
Refer a patient
To refer a patient for breast cancer radiotherapy at GenesisCare, complete our referral form or make an enquiry.
Skin cancerisation radiotherapy
Non-melanoma skin cancer (NMSC) is the most common cancer in the UK. Although it can arise in isolation, a significant group of patients have extensive skin field cancerisation (ESFC) where pre-cancerous lesions (actinic keratosis) pre-exist with cancerous lesions – basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). This carries a high risk of recurrence of pre-cancerous and cancerous lesions, requiring repeated treatment over many years. The ongoing risk of skin cancer and the physical and cosmetic impacts of disease and interventions can affect the patients’ quality of life.
The increasing role of radiotherapy in treatment of NMSC and skin field cancerisation
Although new BCC or SCCs tend to be treated focally, a field-directed approach is optimal for patients who have substantial field UV damage with multiple AKs. Radiotherapy treatment is indicated for moderate to severe degrees of skin cancerisation where topical treatments are ineffective or intolerable in managing ESFC. It may also be used when multiple surgical excisions have made the patient surgically ‘weary’.
Recently, modern radiotherapy techniques such as Volumetric Modulated Arc Therapy (VMAT) have been used in countries, including Australia, for management of ESFC with positive outcomes. This highly conformal technique gives a more homogenous dose delivery and effectively treats large areas of skin while sparing deep tissue from toxicity, even in highly curved areas, such as whole arms or legs. It can also be used where the skin surface overlies organs that could be harmed by radiotherapy, for example, the scalp while sparing the brain tissue from irradiation.
Based on the wide experience and success of this modality by our parent company in Australia, GenesisCare has introduced VMAT to the UK for the treatment of ESFC. We are the first provider in the UK to offer this treatment as part of our evidence-based approach to introducing advanced and innovative treatments.
Evidence based care
In Australia, more than 4000 patients have been treated with radiotherapy for ESFC and a registry has been established to monitor the safety and efficacy outcomes.
A retrospective study of 41 patients with a total of 63 treatment fields for ESFC and 12 months after radiotherapy found that 86% of patients showed complete clearance of fields after their treatment and no cases of invasive cancers were observed.1 However, eight new skin lesions were identified in seven fields, seven cases of Actinic Keratosis and one of Bowen’s disease. Additionally, 98% of patients graded cosmesis as “excellent” or ”good”.
- GenesisCare, data on file
Patients eligible for VMAT
The following patients can be considered for VMAT radiotherapy treatment at GenesisCare for ESFC where:
- The patient has tried other treatments but they have not prevented recurrence of further pre-cancerous lesions (AK)
- The patient has tried other treatments but they have not prevented multiple skin cancer formation
- The patient is immunosuppressed (e.g. due to solid organ transplantation)
- The patient has not tolerated other treatments
Governance
The VMAT skin service has been developed in collaboration with our parent company in Australia, where more than 4,000 patients have been treated. Protocols are reviewed by our UK Clinical Governance Committee and are delivered through the Skin Clinical Reference Group, selected for their expertise and experience of skin radiotherapy treatments.
Partner with us
We are always looking to collaborate with talented consultants.
As a dermatologist, plastic surgeon or related skin cancer specialist, we invite you to partner with us. We welcome requests from individuals and academic institutions to access our state-of-the-art equipment and clinical support for treatment and research. You can also build your knowledge and network with peers at our many CPD events.
Make an enquiry
Please make an enquiry for further information about partnering with us.
Rectal spacers
We design our care to help give patients the best possible life outcomes. Which is why we offer rectal spacers as part of our standard pre‑radiotherapy treatment.
Rectal spacers are a game changer in reducing the effect of toxicity to normal tissues such as the bowel. The spacer is inserted between the rectum and the prostate to temporarily move the anterior rectal wall further away from the prostate. The aim is to reduce the amount of radiation delivered to the rectum and limit toxicity, therefore reducing bowel, urinary and sexual function related side effects.
The rectal spacer lasts for the entire course of prostate radiotherapy treatment and is completely absorbed by the patient’s body over time.
How do rectal spacers work?
The rectal spacer creates space between the prostate gland and rectum, enabling a more accurately targeted radiation dose to the prostate while avoiding unnecessary radiation to healthy organs and tissue.
What are the benefits?
Studies have shown that using rectal spacers in men undergoing radiotherapy for prostate cancer is safe and effective and has been NICE-approved since 2017.[1] By creating perirectal space, the rectal spacer reduces rectal irradiation and results in fewer patient-reported declines in bowel, urinary and sexual function, and improves quality of life (QOL).[2-7]
How are rectal spacers implanted?
A rectal spacer insertion is a minimally invasive procedure that takes around 30 minutes.
Our urologists use a needle to insert the rectal spacer between the prostate and the rectum using an ultrasound guided transperineal approach. The rectal spacer stays in position during the radiotherapy treatment cycle then naturally biodegrades to be absorbed by the body over time.
The procedure is routinely performed under local anaesthetic at a number of our outpatient centres as a safe and convenient alternative to having the insertion under general anaesthetic within a hospital environment.
We accept referrals for patients receiving prostate radiotherapy treatment within the GenesisCare network, or from patients just wishing to self-fund the rectal spacer insertion.
Insertions are still available as a day case procedure, performed by urologists within the GenesisCare network at a number of our national partner hospitals, located throughout the UK.
References
- National Institute for Health and Care Excellence (2017) Biodegradable spacer insertion to reduce rectal toxicity during radiotherapy for prostate cancer (Interventional procedures guidance [IPG590]). Available at: https://www.nice.org.uk/guidance/ipg590 [Accessed 14 October 2020]
- Mariados N, Sylvester J et al (2015). Hydrogel Spacer Prospective Multicenter Randomized Controlled Pivotal Trial: Dosimetric and Clinical Effects of Perirectal Spacer Application in Men Undergoing Prostate Image Guided Intensity Modulated Radiation Therapy. International Journal of Radiation Oncology Biology Physics.92 (5) 971-977.
- Uhl M, Herfarth K et al (2014). Absorbable hydrogel spacer use in men undergoing prostate cancer radiotherapy: 12 month toxicity and proctoscopy results of a prospective multicenter phase II trial. Radiation oncology 9:96. Song DY, Herfarth KK et al (2013). A multi-institutional clinical trial of rectal dose reduction via injected polyethylene-glycol hydrogel during intensity modulated radiation therapy for prostate cancer: Analysis of dosimetric outcomes. International Journal of Radiation Oncology Biology Physics.87 (1) 81-87.
- Wolf F, Gaisberger C et al (2015). Comparison of two different rectal spacers in prostate cancer external beam radiotherapy in terms of rectal sparing and volume consistency. Radiotherapy & Oncology 116 (2) 221- 225.
- Gez E, Cytron S et al (2013). Application of an interstitial and biodegradable balloon system for prostate-rectum separation during prostate cancer radiotherapy: a prospective multi-center study. Radiation Oncology 2013, 8:96
- Chapet O, Decullier E et al (2015). Prostate hypofractionated radiation therapy with injection of hyaluronic acid: Acute toxicities in a phase 2 study. International Journal of Radiation Oncology Biology Physics.91 (4) 730-736.
- Noyes WR, Hosford CC et al (2012). Human collagen injections to reduce rectal dose during radiotherapy. International Journal of Radiation Oncology Biology Physics. 82: 1918-1922.